"Game changer" rapid genetic test will save hearing of hundreds of babies annually in the UK alone
antibiotics
Occasionally the spreading news is good, and this is good: a 25-minute bedside test that will determine if newborns in need of antibiotic treatment have a gene that could result in permanent hearing loss if treatment is adminstered with the common emergency antibiotic, Gentamicin.
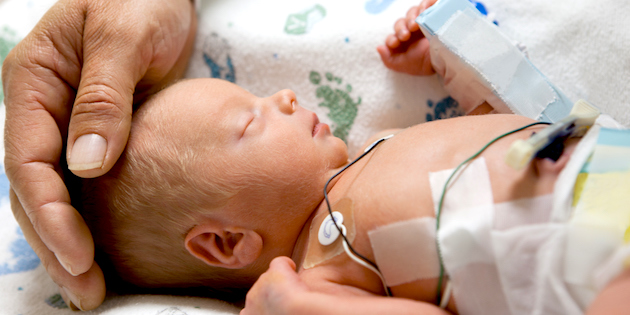
Until now – and successful trials by the UK's National Health Service (NHS) – the process of searching for the gene possessed by one in 500 babies takes days to perform, too long to be of use because babies admitted to intensive care and needing antibiotics for infections are standardly given the antibiotic Gentamicin within 60 minutes. And then it's too late for the genetically vulnerable.
The thrill that a new test can determine in just 25 minutes whether a baby is vulnerable has been spreading throughout the medical and research community since the publication in JAMA Pediatrics on March 21 of a study showing that MT-RNR1 POCT, this great new kid on the block, meets the objective its developers had in mind at its conception: point-of-care genotyping in the acute neonatal setting to avoid aminoglycoside-induced ototoxicity without disrupting normal standards of care.
It is estimated the test could save the hearing of 180 babies in England alone every year, while also knocking £5m off the annual NHS spend by reducing the need for other interventions, such as cochlear implants. Worldwide, the impact will save the hearing of tens of thousands of babies.
Mike Hobday, Director of Policy and Campaigns at the National Deaf Children’s Society, said: “Up to now the genetic test has taken too long to return from the lab to be useful for babies requiring urgent treatment but a rapid test will be a game-changer.”
Manchester, again in a key role
Trialled by the NHS, and funded by £900,000 from the National Institute for Health Research (NIHR), with support from the charity Royal National Institute for Deaf People, this Genedrive System, as it was christened, is a development by Genedrive, a start-up based at the University of Manchester.
Professor William Newman, who led his team at Manchester Biomedical Research Centre, said the arrival of this five-year project at the point of trial success was "fantastic news for the hundreds of babies – and their parents – who would otherwise lose their hearing when given this common antibiotic in intensive care situations".
“The trial demonstrated that you can deploy rapid genetic testing in a clinical setting, and that the tests can be carried out within the ‘golden hour’ when severely unwell babies should be treated with antibiotics,” said Prof. Newman.
Fast implementation in UK hospitals
The University of Manchester has confirmed that following the completion of the study, the NHS Genomic Medicine Service Alliance and the NHS "will be exploring how this technology can be launched as part of a clinical service through the NHS Genomic Medicine Service." Training is under way for around 300 nurses to use the bedside machine across the Manchester University NHS Foundation Trust at Saint Mary’s Hospital, Wythenshawe Hospital, and North Manchester General Hospital. Use is expected to be routine just weeks after the publication of the trial's success.
Genomic medicine is transforming healthcare
David Budd, CEO of Genedrive, said: “There is a significant drive within the NHS to alert healthcare professionals to the impact of antibiotic-induced hearing loss and encourage them to consider genetic testing prior to initiation of treatment. It’s a great example of using human genetics to guide specific therapy, which is now taking front and centre in clinical management globally. The application of Genedrive’s technology shows how a rapid, affordable, point-of-care test could impact patients’ treatment and quality of life across this as well as a wide range of fields.”
While the Chief Scientific Officer for England and Senior Responsible Officer for Genomics in the NHS, Professor Dame Sue Hill, underlined that “Genomic medicine is transforming healthcare, and this is a powerful example of how genetic testing can now be done extremely quickly and become a vital part of triage – not only in intensive care but across our services.
Source: University of Manchester News
 Sign in
Sign in

